University Policies and Procedures Manual (previously Business Policies and Procedures Manual)
Controlled Substances
UPPM 45.75
For more information contact:
Office of the Campus Veterinarian
509-335-6246
Forms:
Policy
This policy addresses obtaining, using, storing, recordkeeping, dispensing, and disposing of controlled substances at WSU. This policy provides information and procedures to enable individuals and departments to comply with state and federal law and to meet applicable safety standards.
This policy does not supersede state or federal law. In the case of any inconsistency, state and federal law govern.
Note: This policy does not apply to licensed pharmacies or pharmacists at WSU. Any entity or individual licensed as a pharmacy or pharmacist must follow all laws, rules, and regulations that pertain to that license.
Important! Improper storage, recordkeeping, dispensing, or disposal of controlled substances may result in civil and criminal liability, which may include incarceration, or license suspension or revocation.
This policy includes the following subsections:
- DEA Criteria
- Registration
- Screening Employees
- Ordering
- Recordkeeping Requirements
- Security
- Disposal
- Abandoned Controlled Substances
DEA Criteria
Controlled substances are drugs or other chemicals that have the potential to be addictive or habit forming. The U.S. Drug Enforcement Administration (DEA) has separated controlled substances into five schedules based on potential to be habit forming and usefulness in medicine. The comprehensive DEA schedule is available on the DEA website.
Below are the criteria established by the DEA for narcotic and nonnarcotic controlled substances.
Schedule I
Schedule I substances are defined as drugs or other substances that have:
- A high potential for abuse;
- No currently accepted medical use in the United States; and
- A lack of accepted safety for use under medical supervision.
Examples of Schedule I substances include: heroin; marijuana.
Schedule II
Schedule II substances are defined as drugs or other substances that:
- Have a high potential for abuse;
- Currently have an accepted medical use in the United States, or have a currently accepted medical use with severe restrictions; and
- If abused may lead to severe psychological or physical dependence.
Examples of Schedule II substances include: methamphetamine; pentobarbital.
Schedule III
Schedule III substances are defined as drugs or other substances that:
- Have a potential for abuse less than Schedule I or II substances;
- Currently have an accepted medical use in the United States; and
- If abused may lead to moderate physical and psychological dependence.
Examples of Schedule III substances include: beuthanasia solution; ketamine; testosterone.
Schedule IV
Schedule IV substances are defined as drugs or other substances that:
- Have a potential for abuse less than Schedule III substances;
- Currently have an accepted medical use in the United States; and
- If abused may lead to low physical or psychological dependence.
Examples of Schedule IV substances include: Valium; butorphanol.
Schedule V
Schedule V substances are defined as drugs or other substances that:
- Have a potential for abuse less than Schedule IV substances;
- Currently have an accepted medical use in the United States; and
- If abused may lead to limited physical or psychological dependence.
Examples of Schedule V substances include: pregabalin; not more than 100mg of opium per 100ml, or per 100g.
Responsibilities
Every person who engages in research with controlled substances must be registered with the DEA and the Washington State Board of Pharmacy or be an authorized user under an existing registrant.
Registrants are responsible for:
- Complying with federal and state laws.
Note: State laws may be more restrictive than federal laws. The stricter law applies.
- Ensuring that each employee who works with controlled substances is qualified under federal regulations. (21 CFR 1301.90) Completion of an employee questionnaire is required (see Screening Employees).
- Ordering and receiving controlled substances purchased under the registrant’s license (see Ordering Substances).
- Ensuring that controlled substances are securely stored to protect the chemicals from theft or misuse (see Storage).
- Maintaining proper recordkeeping for controlled substances, including inventory and tracking (see Recordkeeping Requirements).
- Reporting any theft or loss of controlled substances (see Theft or Loss).
- Properly disposing of controlled substances (see Disposal).
Registration
Controlled substance use in Washington requires participating individuals and/or departments to register with both the federal Drug Enforcement Administration (DEA) and the Washington State Department of Health Board of Pharmacy.
Note: DEA registration requires prior registration with the Washington State Department of Health Board of Pharmacy.
Washington State Department of Health (DOH) Board of Pharmacy Registration
Prior to registering with the DEA, applicants in the state of Washington must obtain controlled substance researcher registration from the Washington State Department of Health (DOH) Board of Pharmacy. There is an annual registration fee. The state fee may not be waived. The current fee schedule is available from the DOH Pharmacy Professional Licensing Information website.
Different registrations are required for different activities involving controlled substances. Further, one registration type may not cover two different activities, such as research with controlled substances and dispensing of controlled substances. The required forms and detailed instructions are available on the DOH Pharmacy Professional Licensing Information website.
Upon completion and approval of the state registration, the Board of Pharmacy issues a state license and number to the applicant. The applicant must include the state license number on the DEA registration application.
DEA Registration
As with state registration, different activities may require different DEA registration types.
Forms
The required forms and detailed instructions are available from the DEA Forms and Applications website.
Research or Laboratory Chemical Analysis
To conduct research on, or with, controlled substances in Schedules II-V or to conduct analysis with controlled substances listed in any schedule, applicants complete:
- DEA Form 225: New Application for Manufacturer, Distributor, Researcher, Analytical Laboratory, Importer, Exporter; or
- DEA Form 225A: Renewal Application for Manufacturer, Distributor, Researcher, Analytical Laboratory, Importer, Exporter.
Dispensing Controlled Substances and/or Instructional Activity
To obtain authorization to dispense controlled substances on any schedule for clinical and hospital use (including clinical research) or to conduct instructional activity only with Schedule II-V controlled substances, applicants complete:
- DEA Form 224: New Application for Retail Pharmacy, Hospital/Clinic, Practitioner, Teaching Institution, or Mid-Level Practitioner; or
- DEA Form 224A: Renewal Application for Retail Pharmacy, Hospital/Clinic, Practitioner, Teaching Institution, or Mid-Level Practitioner.
Required Information
On all DEA registration forms, University applicants must:
- Check the Request Fee Waiver box to indicate eligibility to waive the annual DEA registration cost.
An employee of a federal or state agency, e.g., WSU, who is required to obtain DEA registration to carry out their duties is exempt from DEA registration fees.
- Provide the following investigator information:
- Name
- Physical address
- Mailing address
- Institution
- State license number
- A qualifications statement including a curriculum vita with bibliography for each investigator in the research project.
- Provide the following project information:
- Title
- Statement of purpose
- Controlled substance name
- Amount of controlled substance needed
- Location of research
- Security statement
- Technical description of the substance use
Screening Employees
Authorized users (i.e., designated employees) of the registrant (e.g., principal investigator (PI)) may engage in approved activities under the direction of the registrant. Approved activities may include:
- Access to the controlled substances storage unit.
Such access is defined as use, control, or possession of a key to the storage unit or the means to unlock the storage unit.
- Administration of controlled substances.
- Destruction of controlled substances.
The registrant must screen potential authorized users prior to assigning work with controlled substances (see Authorized User Questionnaire).
Criminal Records
DEA recommends that registrants conduct inquiries concerning employees’ criminal records.
Authorized User Questionnaire
Before allowing an authorized user access to controlled substances, the registrant must require the applicant to complete a questionnaire as part of the screening process.
Important! The questionnaire is mandated and providing access to persons who would answer “yes” to any of the questions is a violation of federal law. (21 CFR 1301.90)
Questions on the questionnaire are as follows:
- Have you been convicted of a felony within the past five years or any misdemeanor within the past two years (excluding traffic, juvenile, and military offenses, unless under a general court-martial), or are you presently charged with committing a criminal offense?
- In the past three years, have you knowingly used narcotics, amphetamines, or barbiturates other than those prescribed to you by a physician?
- Have you ever had an application for DEA registration denied or had DEA registration revoked?
The registrant must have each employee who is authorized by the registrant to handle DEA controlled substances under the registrant’s supervision complete a questionnaire. The registrant must have each new hire complete a questionnaire before they are allowed to handle DEA controlled substances.
The registrant must keep completed questionnaires on file at the registered location, as per the state and DEA registration forms.
Ordering Substances
Schedule I or II
Any person registered to conduct research or otherwise work with Schedule I or II controlled substances must use DEA Form 222 (Official Order Form) to place an order for such chemicals.
Note: See additional procedures under Schedule I Substance That is Not Commercially Available on regarding orders for Schedule I controlled substances that are not commercially available.
Note: The registrant must personally order and receive any Schedule I or II controlled substances. The registrant may not delegate this duty to another employee. To place an order:
- The registrant contacts a drug supplier regarding ordering a commercially-available Schedule I controlled substance or a Schedule II controlled substance.
- The registrant completes a DEA Form 222 and faxes it to the supplier.
- After receiving the completed form, the supplier prepares to fill the order.
- The registrant processes payment for the order with a Create and Change Purchase Order or a Create Requisition business process in Workday. See UPPM 70.10 and the Workday reference guides (Create and Change Purchase Order Create Requisition) for instructions. Registrants are not to purchase controlled substances with procurement cards (see UPPM 70.08.)
Additional Instructions
Further instructions regarding completing DEA Form 222 are available in 21 CFR 1305-13.
Schedule I Substance That is Not Commercially Available
To obtain a Schedule I controlled substance that is not commercially available, the registrant must submit a request to the National Institute on Drug Abuse (NIDA); by telephone to 301-443-1124; or through the National Institute on Drug Abuse website.
Schedule III – V
University registrants may order Schedule III-V controlled substances using WSU’s normal purchasing procedures and documentation.
In accordance with University policies and procedures, registrants may use Create and Change Purchase Order or a Create Requisition business processes in Workday to place orders for Schedule III-V controlled substances. See UPPM 70.10 and the Workday reference guides (Create and Change Purchase Order Create Requisition) for instructions. Registrants are not to purchase controlled substances with procurement cards (see UPPM 70.08.)
Recordkeeping Requirements
The following records should be maintained at the registrant’s location (as identified on the registration).
- Employee screening and authorization records (see Screening Employees).
- Completed order forms (see Ordering Substances).
- Inventory records (see Inventory Procedures ).
- Controlled substance tracking records (including drug dispensing records) See Controlled Substance Tracking.
If the registrant wishes to maintain controlled substance records at a central location other than the registered location, he or she must send a notification to the DEA. Requirements regarding the information that must be submitted in the notification is available in the Code of Federal Regulations, 21 CFR 1304.04.
Retention Requirements
For minimum retention requirements for controlled substance records, see the All-University Records Retention Schedule: Safety Records table on UPPM 90.01.
For further information regarding University records retention and disposition, see UPPM 90.01. For minimum retention requirements for other applicable University records, refer to the other All-University Records Retention Schedule tables at the end of UPPM 90.01.
Controlled Substance Tracking
Registrants must maintain the following in a separate, bound and sequentially numbered tracking document for each controlled substance. Each tracking document must include:
- Records tracking each container of controlled substance as well as records that detail each time a controlled substance is used.
- Dispensing records accounting for all of the controlled substance in milliliter (ml) or milligram (mg) units.
- An entry for each time any material was removed from the container, including any quantity wasted or disposed, and initials of the user.
The registrant must maintain records for Schedule I and II controlled substances separately from records for Schedule III-V controlled substances. See the sample controlled substance tracking record in Fig. 1.
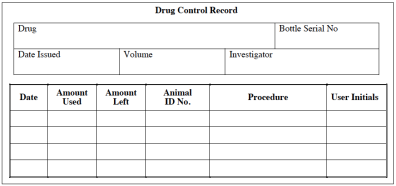
Important! It is a felony for a registrant to provide a controlled substance to a person who is not registered with the DEA or is not one of the registrant’s authorized users. A transfer of controlled substances may occur between two DEA registrants only. A transfer of Schedule I or II controlled substances must be accompanied by a DEA Form 222 (Official Order Form) completed by the registrant receiving the substances.
Inventory Procedures
Each registrant must complete an inventory annually as required by the Washington Board of Pharmacy. The registrant must:
- Maintain the inventory record at the registered location (unless she or he sends a notification to DEA that such records are maintained at a specified central location).
- Retain and make the inventory record available in accordance with University records retention requirements (see Recordkeeping Requirements above).
- Update the inventory record on the effective date of a rule (from the DEA) that adds a substance to the list of controlled substances, Schedules I-V.
Inventory Record Form
Complete and/or print copies of the Controlled Substance Inventory Record as needed.
Required Inventory Data
The inventory is to include the following information, based upon the types of controlled substances maintained.
Commercial Purchased
For commercially-purchased controlled substances, i.e., controlled substances obtained in a finished form, the inventory records must include the following:
- Registrant’s name, physical address, mailing address, and DEA registration number.
- Date the inventory was taken.
- Name of each substance.
- Form of each substance (i.e., 10-milligram tablet, or 10-milligram concentration per fluid ounce, or milliliter).
- Number of units or volume of each commercial container (i.e., 100-tablet bottle or 3-milliliter vial).
- Exact count of dosage units.
- Number of commercial containers of each substance form.
- Registrant signature and date signed.
Note: Schedule I and II substances must be separated from other substances on the inventory.
Damaged, Defective, Impure, or Not Commercially Purchased
For damaged, defective, or impure substances, and/or substances that were not commercially purchased, the inventory records must include an exact count of the dosage units or the container must be graduated to reflect its content. Such inventory records must also include the following:
- Registrant’s name, address, and DEA registration number.
- Date the inventory was taken.
- Name of substance.
- Total volume of substance or total number of units (i.e., 50 10mg tablets).
- Reason that the registrant (e.g., researcher) is maintaining the substance.
- Registrant’s signature and date signed.
Security
Theft or Loss
If there is a theft or loss of controlled substances, the registrant must notify the Washington State University Police (or the local police department, if no University Police department is available at the location). The notification must occur as soon as the theft or loss is discovered.
The registrant must notify the area DEA Field Office in writing within one business day of the discovery of the theft or loss. The registrant must also complete and submit a DEA Form 106 (available from the DEA Theft/Loss Reporting website) to the area DEA Field Office and send a copy of the completed form to the Washington State Board of Pharmacy. (21 CFR 1301.76; WAC 246-887-020)
Storage
Storage locations must be consistent with the locations identified on the registrant licenses. Registrants are generally permitted to keep controlled substances in substantially constructed, double-locked cabinets or safes, as described below. Narcotics cabinets are strongly recommended, to ensure that the registrants’ storage complies with regulations.
Important! It is unacceptable for a registrant to leave the keys to the cabinet in the laboratory. Keys allowing access to controlled substances must be in the possession of the registrant.
Schedules III-V
For Schedules III-V, registrants must keep controlled substances in a substantially constructed, double-locked cabinet or safe.
Schedules I and II
For Schedules I and II, registrants must keep the controlled substances in a substantially constructed, double-locked cabinet that cannot be easily forced open, with the cabinet secured to a wall or otherwise not removable. For specific information regarding physical security requirements, see 21 CFR 1301.71 to 1301.76.
Disposal
When a DEA registrant has a controlled substance that is expired or unwanted, the registrant must contact Environmental Health and Safety (EH&S) at WSU Pullman for the disposal of the substance. WSU Pullman EH&S is the University’s registered DEA reverse distributor (see also SPPM 5.66).
EH&S disposes of the controlled substances using DEA- and DOH-approved procedures. For additional assistance or questions about controlled substance disposal, contact EH&S; telephone 509-335-3041.
Abandoned Controlled Substances
Under no circumstances is a DEA registrant to abandon controlled substances.
Faculty are to follow the procedures in UPPM 60.38 prior to termination of employment to ensure that materials are properly disposed of or transferred.
If controlled substances are abandoned, contact the Office of the Campus Veterinarian for assistance. Substances are to be disposed or transferred. The Office of the Campus Veterinarian determines the applicable procedures on a case-by-case basis.
_______________________
Revisions: Nov. 2024 (editorial); Jan. 2021 (Rev. 560); Dec. 2013 – new policy (Rev. 423).