Safety Policies and Procedures Manual
Research Biosafety Policy
SPPM 4.20
For more information contact:
Office of Research Assurances
509-335-7195
Policy
All possession, use for research purposes, and transportation of biological and select agents hazardous to human, animal, and plant health must conform to regulations and guidelines established by federal, state, and local agencies.
Biological and select agents used in research purposes in accordance with this policy include infectious organisms, recombinant or synthetic nucleic acid (r/sNA) molecules, oncogenic viruses, viroids, prions, and transgenic organisms. See Biohazardous Agents.
Public Health and Workplace Biosafety
The following SPPM sections provide additional policies and procedures regarding public health and workplace biosafety issues:
- Bloodborne pathogens (see SPPM 2.44)
- Food safety and sanitation (see SPPM 6.20)
- Hantavirus (see SPPM 6.33)
- Bird and bat waste (see SPPM 6.35)
- Drinking water systems (see SPPM 6.50)
- Sewage spills (see SPPM 6.70)
- Biohazard wastes (see SPPM 4.24)
- Hazardous materials shipping (see SPPM 5.40)
Non-Research Clinical Health Service Biosafety
University units, e.g., Cougar Health Services, providing non-research clinical health services must also follow applicable regulations and guidelines established by federal, state, and local agencies. Supervisors in non-research clinical health services units are responsible for training staff accordingly.
Purpose
This research biosafety policy provides faculty, staff, and students at all WSU locations and the surrounding communities with guidelines for managing risks associated with the use or control of biohazardous agents in the research setting.
This policy assists the University, the Institutional Biosafety Committee (IBC), University departments, WSU Extension, and principal investigators involved in research, teaching, and/or diagnostics in identifying and implementing appropriate safeguards.
The WSU Biosafety Officer, the Office of Research Assurances (ORA), and the Office of Research Support and Operations (ORSO), have copies of the federal and state guidelines available for reference.
Definitions
Appropriate Biosafety Levels
Appropriate biosafety levels (BSL) are established in conformance with regulations or guidelines from federal, state, and local agencies including, but not limited to:
- Centers for Disease Control and Prevention (CDC)
- National Institutes of Health (NIH)
- United States Department of Agriculture (USDA)/Animal and Plant Health Inspection Service (APHIS)
Appropriate biosafety levels are set for laboratory research, for research involving animals (ABSL), and for research involving plants (BL-P) for each biohazardous agent and protocol in use.
Biohazardous Agents
The term “biohazardous agents” includes, but is not limited to, all of the following:
- Infectious organisms (e.g., bacteria, chlamydiae, fungi, parasites, prions, rickettsias, and viruses) which can cause disease in humans, animals, or plants.
Prions are infectious proteinaceous particles associated with transmissible spongiform encephalopathies in animals and humans. - Infectious organisms which can cause significant environmental or agricultural impact.
- Materials that may harbor infectious organisms, such as human or primate tissues, fluids, cells, or cell cultures.
- Recombinant or synthetic nucleic acid molecules (r/sNA), including:
- Recombinant nucleic acid molecules (e.g., cloning);
- Synthetic nucleic acid molecules, including those that are chemically or otherwise modified but can base pair with naturally-occurring nucleic acid molecules; and
- Cells, viruses, and organisms, containing such molecules.
- Transgenic organisms (i.e., animals, plants, or microbes which have been modified by genetic engineering).
- Select agents and toxins, in accordance with the Federal Select Agent Program (FSAP). The FSAP includes biological select agents and toxins (BSAT) listed in the Code of Federal Regulations (including, but not limited to, 42 CFR Part 73, 7 CFR Part 331, and 9 CFR Part 121).
BSAT are capable, if released, of generating a serious public health crisis or are high-consequence livestock pathogens. Transfer of BSAT is limited and controlled by the National Centers for Disease Control and Prevention (CDC).
For more information, see the ORA’s Potentially Biohazardous Materials website.
Dual-Use Research of Concern (DURC)
Dual-use research of concern (DURC) is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that can be directly misapplied to pose significant threats. Such threats can have broad potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, material, or national security.
Organizational Structure
In order to ensure that the requirements of the research biosafety program are met, a collective and interactive effort between various University organizations, departments, and individuals at all University campuses is required. The chart below (Figure 1) illustrates the current University organizational structure and departments, units, and committees that direct and/or support the research biosafety program. It is intended that the administrators, principal investigators, and employees at each campus work in cooperation with the specified safety groups to support the University research biosafety program.
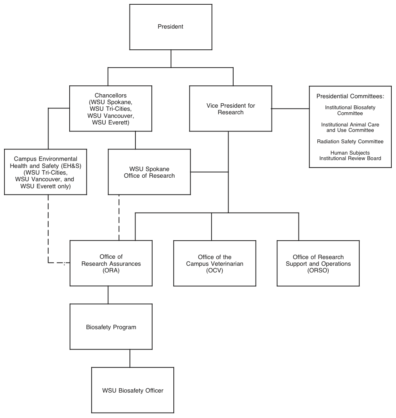
Responsibility
The following departments collaboratively work to maintain a comprehensive, integrated research biosafety program:
- The Office of Research Assurances (ORA), including the WSU Biosafety Officer (BSO);
- The WSU Tri-Cities, WSU Vancouver, and WSU Everett campus offices of Environmental Health and Safety (EH&S), in consultation with ORA;
- The WSU Spokane Office of Research, in consultation with ORA;
- The Office of the Campus Veterinarian (OCV); and
- Elements within ORSO.
The program is intended to ensure that risks associated with the use of biohazardous agents in research settings do not exceed the benefits accrued from the University activities.
The specific elements of the program are governed by a variety of state and federal laws and regulations. Many of the responsible offices coordinate activities with Presidential committees. (See Fig. 1.)
Select Agent Program Administration
The Federal Select Agent Program (FSAP) is administered at each entity by a Responsible Official and Alternate Responsible Official(s). At WSU, the Vice President for Research serves as the Responsible Official. The Responsible Official may name Alternate Responsible Officials including, but not limited to, the Director of the Office of Research Assurances and the BSO. In the case of a change of leadership for the Responsible Official, an Alternate Responsible Official temporarily assumes the role until a new Responsible Official is approved by the President and active.
Office of Research and Finance Chancellors
The Vice President for Research and the WSU Spokane, Tri-Cities, Vancouver, and Everett campus chancellors implement the research biosafety policy. (See Responsibility.)
The Vice President for Research or representative(s) and the chancellors or representatives are jointly responsible to:
- Review the research biosafety policy at least once a year and report University activity, as required, to federal and state agencies.
- Ensure compliance with applicable regulations and guidelines from federal, state, and local agencies.
- Identify funding mechanisms for IBC-recommended biosafety-related capital projects.
Office of Research Assurances (ORA)
The Office of Research Assurances (ORA):
- Assists the Vice President for Research in administering and implementing a comprehensive research biosafety program.
- Ensures that all University investigators, including those at all campuses, research and extension centers (RECs) or stations (RESs), and WSU Extension county offices conduct activities with biohazardous agents at appropriate biosafety levels. Coordinates with the Institutional Animal Care and Use Committee (IACUC) for housing of laboratory animals at appropriate biosafety levels.
Institutional Biosafety Committee (IBC)
The University’s Institutional Biosafety Committee (IBC):
- Reports to the University President through the Vice President for Research regarding compliance with federal and state regulations and biosafety needs.
- Approves all University protocols (i.e., Biosafety Approval Forms (BAFs)) to conduct activities with biohazardous agents or house laboratory animals at appropriate biosafety levels. University protocols include protocols used at all campuses, RECs, RESs, and WSU Extension county offices. (See the definition of appropriate biosafety levels.)
IBC approval to conduct research is required regardless of funding source.
To obtain copies of the BAF and instructions, select the Forms and Templates link on the Biosafety website.
- Reviews research protocols for compliance with standards.
- Reviews health surveillance measures.
- Reviews all r/sNA research conducted at WSU for compliance with guidelines established by the National Institutes of Health (NIH).
- Recommends assessment of biosafety facilities, procedures, and practices by the BSO and the campus Environmental Health and Safety EH&S departments.
- Requires training for personnel involved with biohazardous agent activities.
- Establishes and implements policies for WSU biohazardous agent activities.
- Advises the Vice President for Research in the resolution of conflicts that arise with interpretation of biosafety standards.
- Approves inspection checklists for approval of BSL and ABSL laboratories and procedures.
- Assists in prioritizing and administering minor and major capital funds for biosafety needs.
- The IBC is designated as the University Institutional Review Entity (IRE). When functioning as the IRE, IBC membership must be organized in a manner that complies with federal DURC policy, including identification, reporting, and oversight.
The IBC and the BSO assist the Vice President for Research as the Institutional Contacts for Dual Use Research (ICDUR).
WSU Biosafety Officer (BSO)
The WSU Biological Safety Officer (BSO):
- Develops, directs, and manages the WSU research biosafety program, in compliance with federal, state, and institutional guidelines.
- Serves as a biosafety resource, including providing technical advice on biosafety procedures and monitoring biosafety trends and regulatory changes, for the following University groups:
- Institutional Biosafety Committee (IBC)
- Institutional Animal Care and Use Committee (IACUC)
- Laboratory Health and Safety Committee)
- Human Subjects Institutional Review Board (IRB)
- Non-Pullman campus EH&S departments
- WSU Spokane Office of Research
- Campus facilities departments
- Hazardous Materials Shipping Program
- Radiation Safety Committee (RSC)
- Assists research investigators and staff with all biosafety issues including the following:
- Interpretation of regulations
- Biocontainment practices
- Biosafety risk assessment
- Development of standard operating procedures (SOPs)
- Development of the laboratory biosafety manuals
- Use of biocontainment equipment
- Assists principal investigators (PIs) regarding management and safety of transgenic organisms.
- Reviews biosafety manuals and standard operating procedures (SOPs) for biohazardous agents. Coordinates with each campus regarding specific details for laboratory approval and inspection when warranted by the nature of a biohazardous agent.
- Develops and maintains the database of biohazardous agents at WSU. The database includes a current inventory of select agents on all University campuses in compliance with 42 CFR Part 73 and Public Law 107-188, “Public Health Security and Bioterrorism Preparedness and Response Act of 2002.”
- Develops and maintains the biosafety cabinet certification program for the University, including inspection of biosafety cabinets and maintenance of a biosafety cabinet database. See SPPM 4.22.
- Acts as a resource when questions arise regarding appropriate decontamination of facilities and equipment.
- Develops, implements, and maintains biosafety training and educational programs for academics, investigators, and staff. This training is in addition to the bloodborne pathogen training provided by the campus EH&S departments.
- Provides advice about response plans in coordination with the affected WSU units’ emergency response plans and the PIs.
- Serves as a resource to provide biosafety consultation to incident commanders (i.e., WSU Police Services, and/or local police and fire services) during emergency responses.
- Coordinates with the campus facilities operations and EH&S departments for corrections, modifications, and/or repairs to physical facilities.
- Coordinates with Facilities Services, Capital on planning and design criteria for major capital projects.
- Provides advice about biohazard waste management in coordination with the campus facilities departments.
- Coordinates with the WSU Tri-Cities, WSU Vancouver, and WSU Everett campus EH&S departments and the WSU Spokane Office of Research to provide biosafety resources and services. Such services include initial and regular periodic inspections of physical facilities and containment equipment for compliance with general CDC, NIH, USDA, and other federal agency guidelines.
Inspections must be conducted in accordance with laboratory inspection checklists developed in coordination with the Biological Safety Officer (BSO) and the IBC. - Provides general guidance about health and safety standards, and assists the BSO and the IBC in reviewing research proposals.
ORA, Central Receiving & Delivery, and Campus Mail and Receiving Departments
ORA hazardous materials shipping specialists, Central Receiving & Delivery, and the campus mail and receiving departments monitor shipments for dangerous goods and hazardous materials (e.g., biohazardous agents) to ensure adherence to the following regulations for all packages shipped from and received at Central Receiving & Delivery and the campus mail and receiving departments:
- Federal Department of Transportation regulations 49 CFR;
- Federal select agent regulations 42 CFR Part 72.6 and Appendix A; and
- International Air Transport Association (IATA).
If dangerous goods or hazardous materials are deemed present:
- WSU Pullman departments are to contact the Office of Research Assurances.
- WSU Spokane (WSUS) departments are to contact the WSUS Office of Research regarding biohazardous agents. Note: For dangerous or hazardous chemicals, WSUS departments are to contact WSUS EH&S.
- Other non-Pullman campus departments are to contact the applicable trained and certified dangerous goods shipping coordinator. See SPPM 5.40.
Central Receiving & Delivery and the campus mail and receiving departments are not held responsible for dangerous goods shipments picked up directly by carriers from University departments. See SPPM 5.40.
Department Administrators
Departmental administrators supervise their employees, students, and visitors with regard to biological risks. The department chair:
- Reviews laboratory accident reports.
- Takes appropriate steps to avoid accidents.
- Ensures that principal investigators and staff are trained for work with biohazards.
- Ensures that principal investigators maintain records of employee safety training.
- Coordinates the development of emergency management plans.
- Approves control methods. (See also SPPM 2.44 regarding bloodborne pathogen exposure control.)
Principal Investigator (PI)
WSU assigns primary responsibility for enforcing rules and procedures for proper control of biohazardous agents to the PIs and supervisors. These duties include:
- Operating in conformance with all applicable regulations and guidelines from federal, state, local, and University policies.
- Implementing a laboratory biosafety manual including standard operating procedures and emergency procedures for managing biohazardous agents for any such research under the PI’s supervision.
- Training employees in safety practices, correcting work errors, identifying defective working conditions, and developing a positive attitude among employees toward accident prevention.
- Maintaining records of employee safety training.
- Responding to small spills and releases.
- Properly decontaminating equipment and facilities as needed.
- Investigating and reporting each accident or unintentional release, initiating corrective action, and recommending improvements to ensure maximum safety for WSU laboratory workers. The PI also sends a copy of each report to the IBC, BSO, and the campus EH&S department.
- Ensuring that operations involving using or storing select agents are conducted according to 42 CFR Part 73 and the U.S. Patriot Act of 2001. This includes taking steps to ensure that no “restricted person,” as defined by the Patriot Act, has access to select agents under the principal investigator’s control.
- Ensuring compliance with all import, export, and material transport permit requirements. See SPPM 5.40.
Employees
Employees assigned to work with biohazardous materials are responsible for complying with safety rules and procedures. These duties include:
- Reporting each accident or spill resulting in human exposure or breach of containment or any action or condition which may cause such a breach to the PI or supervisor.
- Reporting any relevant medical profile change to the PI who will include the change in the employee’s medical surveillance records.
- Maintaining proficient, safe working techniques and containment practices.
- Attending training sessions and safety orientations as directed.
_______________________
Revisions: Oct. 2019 (Rev. 121); Sept. 2007 (Rev. 70); Sept. 1987 – new policy (Rev. 4).